Abstract
The purpose of this retrospective biomarker study of the Canadian Cancer Trials Group (CCTG) MA.31 randomized phase 3 trial (lapatinib vs trastuzumab) of HER2-positive metastatic breast cancer (MBC) was to evaluate the prognostic and predictive biomarker utility of pretreatment serum programmed death ligand 1 (PD-L1) levels.
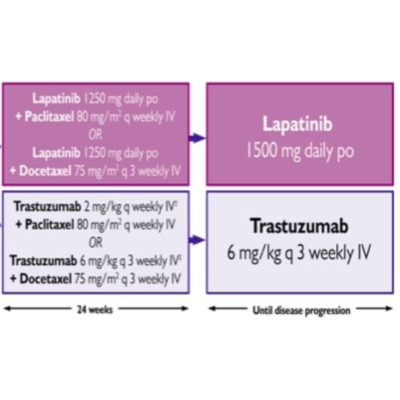
CCTG MA.31 accrued 652 HER2-positive patients; 387 had serum available (185 in the trastuzumab arm and 202 in the lapatinib arm). The Ella immunoassay platform (ProteinSimple, San Jose, California) was used to quantitate serum PD-L1 levels. Stepwise forward Cox multivariable analyses were performed for progression-free survival and overall survival (OS).
In the whole trial population, continuous pretreatment serum PD-L1 levels were not associated with OS. However, within the trastuzumab arm, a higher continuous pretreatment serum PD-L1 level was significant for shorter OS (hazard ratio [HR], 3.85; P = .04), but within the lapatinib arm, pretreatment serum PD-L1 was not associated with OS (P = .37). In the whole trial, in a multivariable analysis for OS, serum PD-L1 (median cut point) remained a significant independent covariate (HR, 2.38; P = .001). There was a significant interaction between treatment arm and continuous serum PD-L1 (bootstrap method; P = .0025): at or above 214.2 pg/mL (the 89th percentile), serum PD-L1 was associated with significantly shorter OS with trastuzumab treatment versus lapatinib treatment.
In the CCTG MA.31 trial, serum PD-L1 was a significant predictive factor: a higher pretreatment serum PD-L1 level was associated with shorter OS with trastuzumab treatment but with longer OS with lapatinib treatment. Immune evasion may decrease the effectiveness of trastuzumab therapy. Further evaluation of elevated serum PD-L1 in advanced breast cancer is warranted to identify patients with HER2-positive MBC who may benefit from novel immune-targeted therapies in addition to trastuzumab.