Abstract
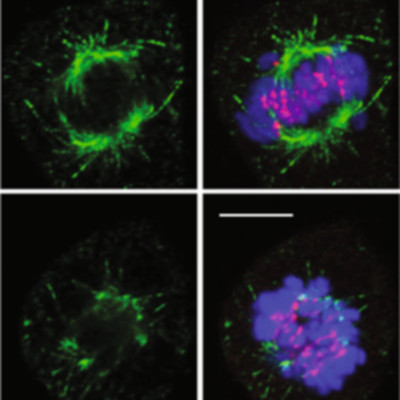
The RNA helicase EIF4A3 regulates the exon junction complex and nonsense-mediated mRNA decay functions in RNA transcript processing. However, a transcriptome-wide network definition of these functions has been lacking, in part due to the lack of suitable pharmacological inhibitors. Here we employ short-duration graded EIF4A3 inhibition using small molecule allosteric inhibitors to define the transcriptome-wide dependencies of EIF4A3. We thus define conserved cellular functions, such as cell cycle control, that are EIF4A3 dependent. We show that EIF4A3-dependent splicing reactions have a distinct genome-wide pattern of associated RNA-binding protein motifs. We also uncover an unanticipated role of EIF4A3 in the biology of RNA stress granules, which sequester and silence the translation of most mRNAs under stress conditions and are implicated in cell survival and tumour progression. We show that stress granule induction and maintenance is suppressed on the inhibition of EIF4A3, in part through EIF4A3-associated regulation of G3BP1 and TIA1 scaffold protein expression.