Abstract
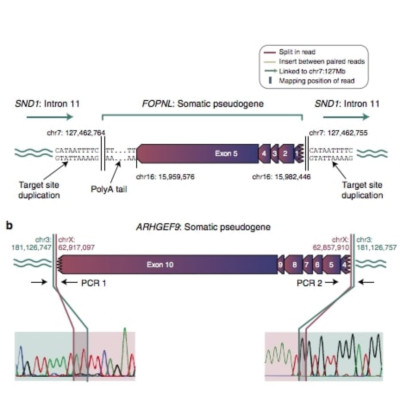
Cancer evolves by mutation, with somatic reactivation of retrotransposons being one such mutational process. Germline retrotransposition can cause processed pseudogenes, but whether this occurs somatically has not been evaluated. Here we screen sequencing data from 660 cancer samples for somatically acquired pseudogenes. We find 42 events in 17 samples, especially non-small cell lung cancer (5/27) and colorectal cancer (2/11). Genomic features mirror those of germline LINE element retrotranspositions, with frequent target-site duplications (67%), consensus TTTTAA sites at insertion points, inverted rearrangements (21%), 5’ truncation (74%) and polyA tails (88%). Transcriptional consequences include expression of pseudogenes from UTRs or introns of target genes. In addition, a somatic pseudogene that integrated into the promoter and first exon of the tumour suppressor gene, MGA, abrogated expression from that allele. Thus, formation of processed pseudogenes represents a new class of mutation occurring during cancer development, with potentially diverse functional consequences depending on genomic context.